1-12 of 22 results
-
Alcohol in fermented foods: Considerations for pregnancy and childhood
Typical health recommendations advise not consuming alcohol during pregnancy or during childhood up to the age of 18. Yet some fermented foods contain alcohol. What does the science reveal about resolving this contradiction? -

Expert Panel at ISAPP Annual Meeting Addresses Probiotic Use for Premature Infants
The use of probiotics in premature infants has been highly topical in recent months. Probiotic use for the prevention of… -

Statistical considerations for the design of randomized, controlled trials for probiotics and prebiotics
By Prof. Daniel Tancredi, UC Davis, USA The best evidence for the efficacy of probiotics or prebiotics generally comes from… -
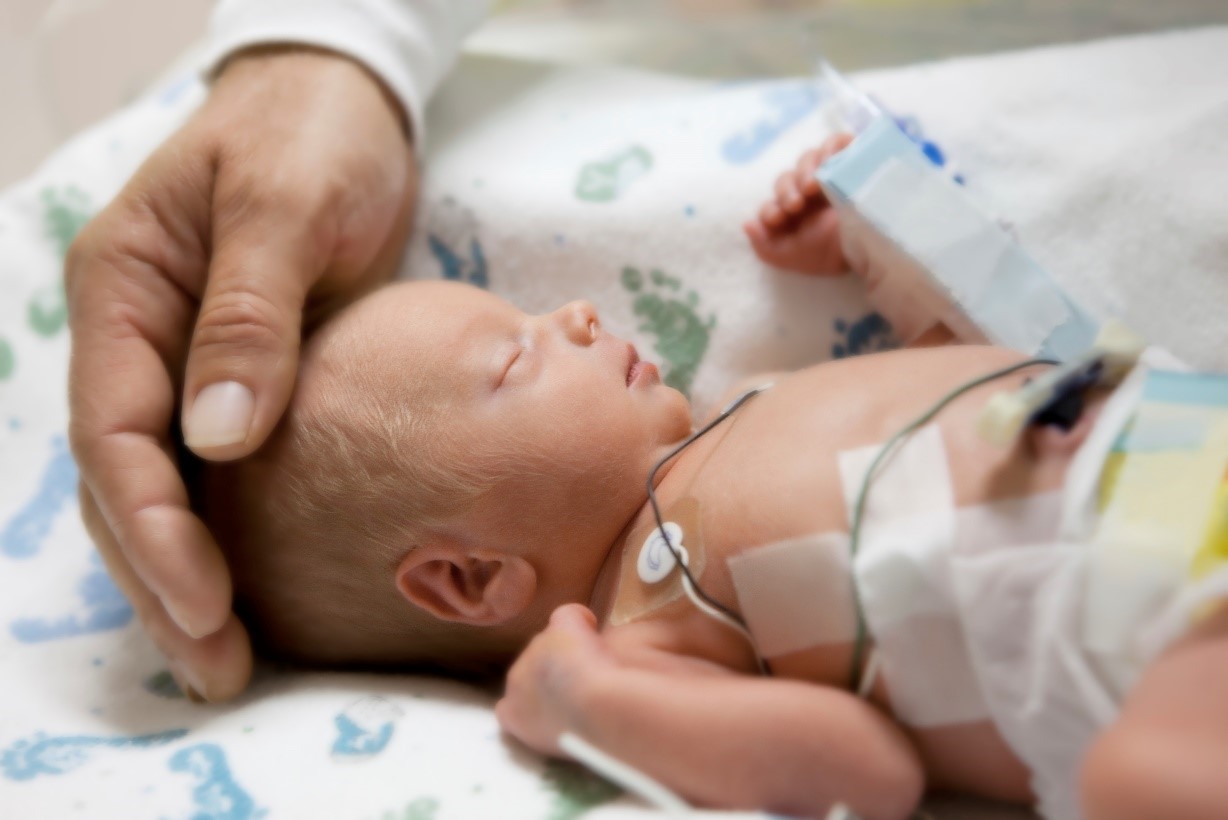
Probiotic Administration in Preterm Infants: Scientific Statement
Board of Directors, International Scientific Association for Probiotics and Prebiotics in collaboration with Dr. Geoffrey Preidis MD PhD, Pediatric Gastroenterology,… -

Are the microbes in fermented foods safe? A microbiologist helps demystify live microbes in foods for consumers
Since very early in my career I was drawn to science communication. I feel that rather than just producing my… -

Can Probiotics Cause Harm? The example of pregnancy
Co-authored by Dr. Maria Carmen Collado, Institute of Agrochemistry and Food Technology-National Research Council (IATA-CSIC), Valencia, Spain Limiting excessive weight… -

Pasteurized Akkermansia muciniphila as a postbiotic: EFSA approval and beyond
By Prof. Seppo Salminen, University of Turku, Finland Earlier this year, the European Food Safety Authority (EFSA) delivered an opinion… -

ISAPP board members give a scientific overview of synbiotics in webinar
Many kinds of products are labeled as synbiotics – but how do they differ from each other? And do they… -

Current issues in probiotic quality: An update for industry
By Dr. Mary Ellen Sanders, ISAPP, Dr. Kit Goldman, USP, Dr. Amy Roe, P&G, Dr. Christina Vegge, Dr. Jean Schoeni,… -

Can dietary supplements be used safely and reliably in vulnerable populations?
By Dr. Greg Leyer, Sr. Director – Scientific Affairs, Chr. Hansen, Inc., Madison, WI and Prof. Dan Merenstein, Department of… -
Follow up from ISAPP webinar – Probiotics, prebiotics, synbiotics, postbiotics and fermented foods: how to implement ISAPP consensus definitions
By Mary Ellen Sanders PhD, Executive Science Officer, ISAPP On the heels of the most recent ISAPP consensus paper –… -
Video Presentation: Behind the scenes of the consensus panel discussion on the definition of fermented foods
Numerous misunderstandings and questions exist around the concept of fermented foods. For example: If a food does not contain live…


