1-6 of 6 results
-
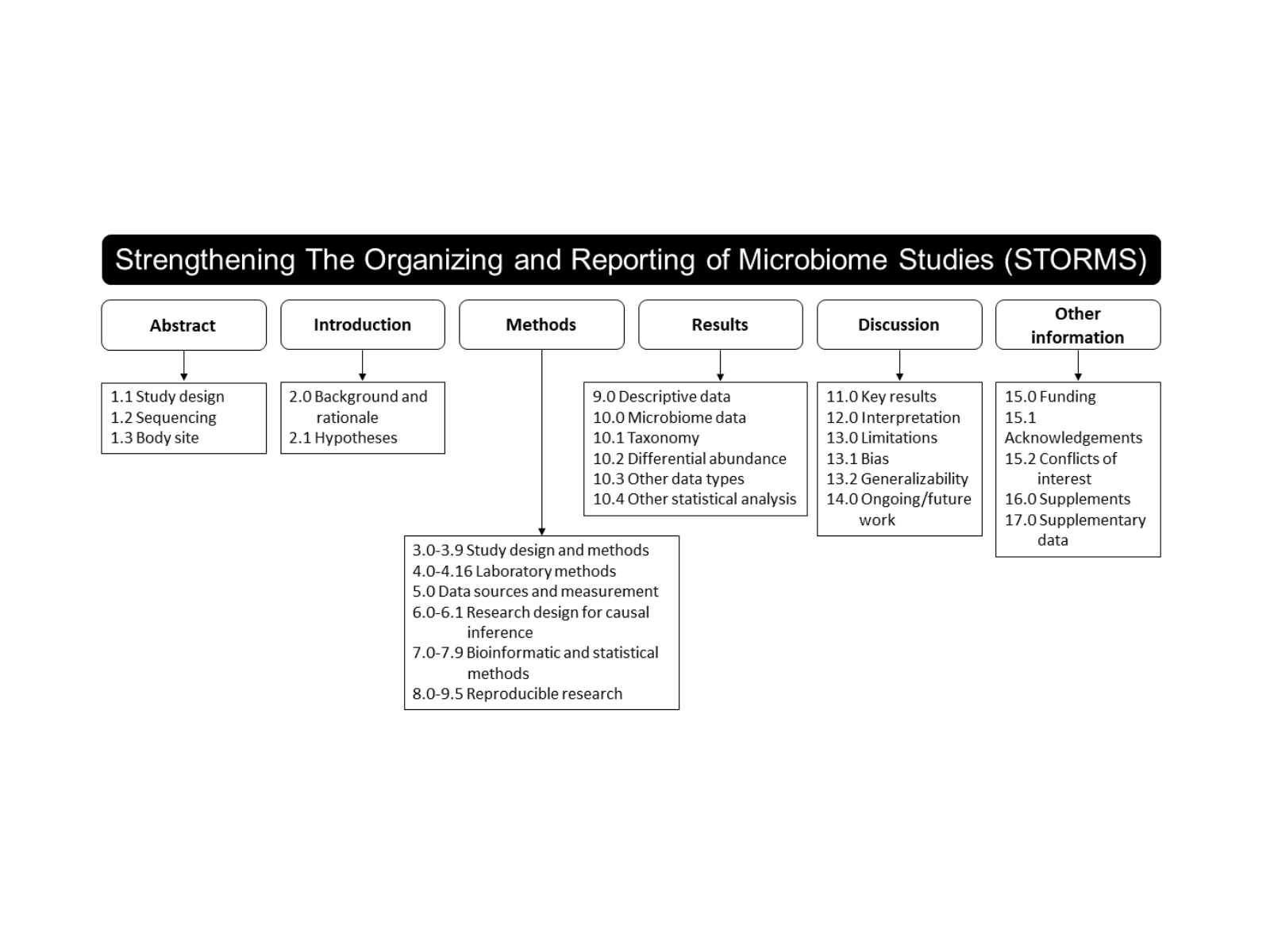
Improving the quality of microbiome studies – STORMS
By Mary Ellen Sanders, PhD, ISAPP Executive Science Officer In mid-March I attended the Gut Microbiota for Health annual meeting…. -
Can dietary supplements be used safely and reliably in vulnerable populations?
By Dr. Greg Leyer, Sr. Director – Scientific Affairs, Chr. Hansen, Inc., Madison, WI and Prof. Dan Merenstein, Department of… -

What’s a Clinician to do When the Probiotic Recommendations from Medical Organizations Do Not Agree?
By Prof. Hania Szajewska, MD, Department of Paediatrics, The Medical University of Warsaw, Poland The scientific literature on probiotics is… -

ISAPP ha estado trabajando en colaboración con la Sociedad de Enterocolitis Necrotizante
La Asociación Científica Internacional para Probióticos y Prebióticos (ISAPP, por sus siglas en inglés), ha estado trabajando en colaboración con… -

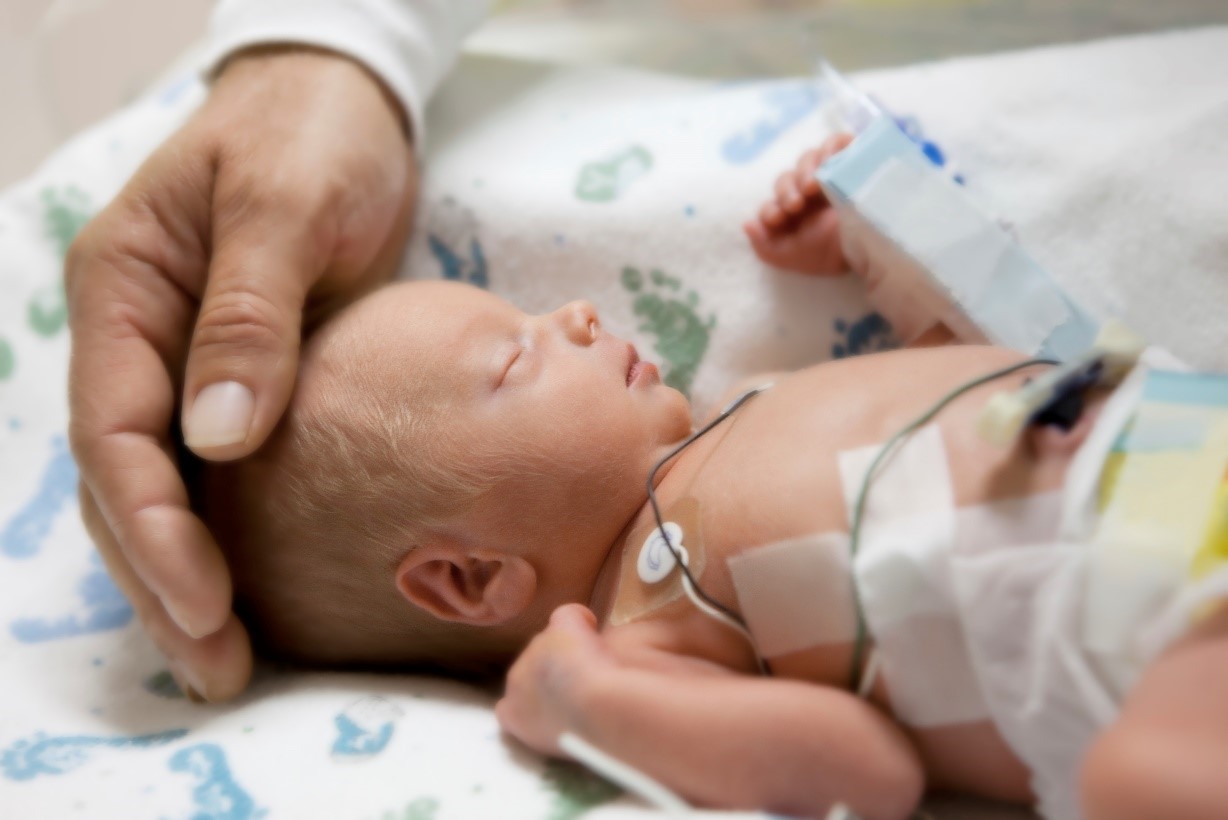
ISAPP collaborates with NEC Society to help parents understand the role of probiotics in reducing the risk of necrotizing enterocolitis
By Dr. Mary Ellen Sanders, PhD, ISAPP Executive Science Officer To date, over 50 clinical trials on probiotics and necrotizing… -

Probiotics to Prevent Necrotizing Enterocolitis: Moving to Evidence-Based Use
By Ravi Mangal Patel, MD, Msc, Associate Professor of Pediatrics, Emory University School of Medicine and Children’s Healthcare of Atlanta….