Probiotics: the importance of the complete product
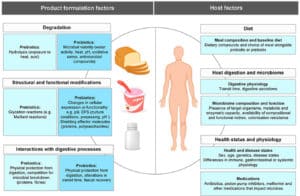
Probiotics are, according to the WHO and later updated by a consensus panel convened by ISAPP, defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host”. Most scientific literature ties probiotic properties to individual strains, although evidence suggests that some health benefits may generalize to the species or genus level. Another important factor in how a probiotic performs is the type of matrix (e.g., a milk drink) that carries the probiotic. Indeed, many successful commercial probiotic products are largely defined by both the probiotic contained and the final product format. A plethora of probiotic products are available, ranging from fermented milks/yogurts, cereal products, juices and freeze-dried products (powders and pills). Some products claim to be probiotic but lack substantiation, such as “probiotic” pizzas and mattresses. It is likely that the probiotic properties are not solely determined by the probiotic strain itself, but also by the harbouring matrix. Hence, in order to fully understand the parameters that drive functionality of a specific probiotic, the total product should be evaluated.
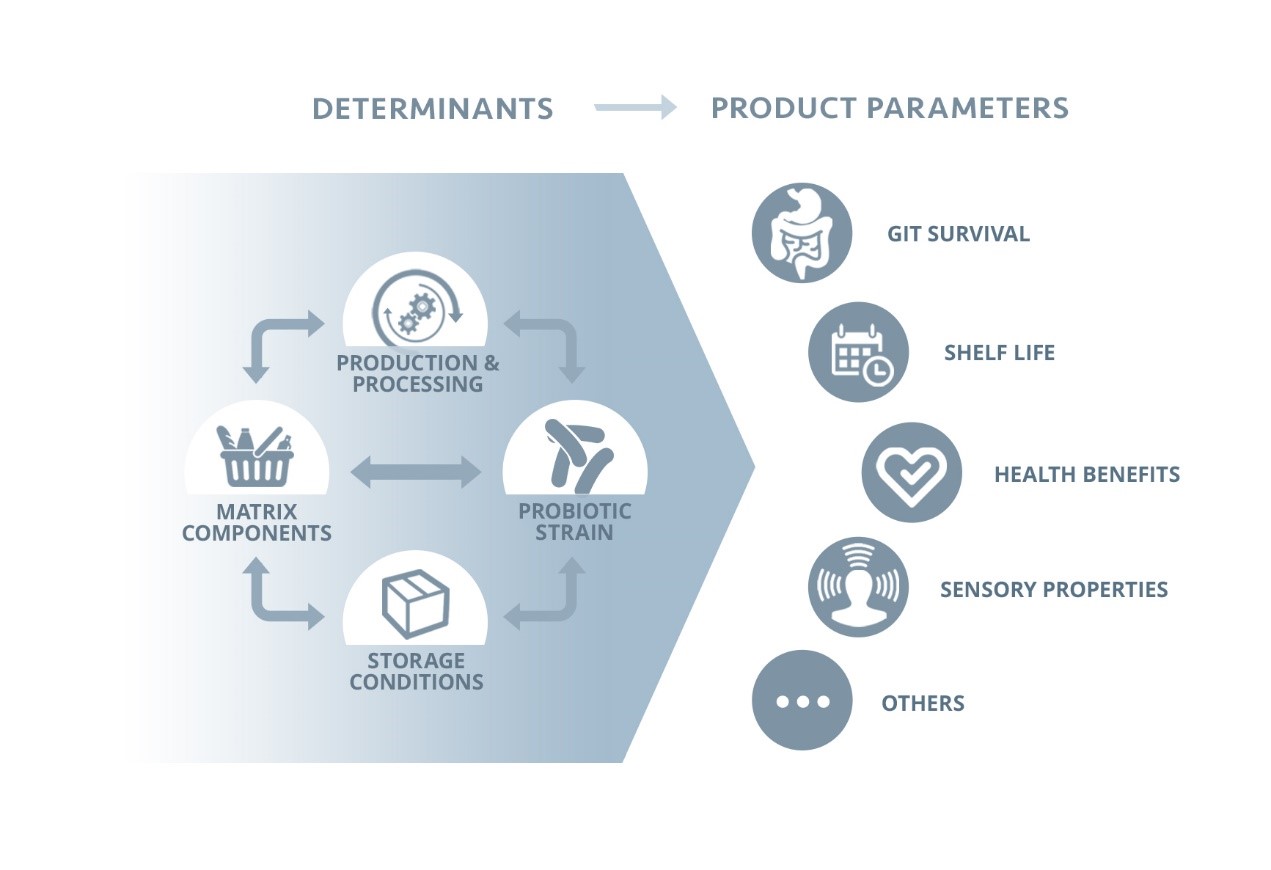
Recently, the influence of the matrix on measures of probiotic functionality was reviewed. The data suggest that the matrix impacts several parameters, including number of viable probiotic microorganisms present in the product through shelf life and survival of the probiotic through the gastrointestinal tract. As an example, the number of viable microorganisms in the product as a function storage time can be profoundly different depending on the combination of probiotic strains and matrices used. Some products in which lyophilized probiotics are incorporated into a peanut butter matrix can have storage times up to 50 weeks. Whey proteins present in milk may improve gastrointestinal tract survival. Therefore, one should be aware that it is likely that viability of the probiotic will be impacted by the carrier matrix.
Another way that matrix can be important is through delivery of additional beneficial substances. For example, milk products contain various vitamins, calcium and high quality protein. In the case of a fermented probiotic product, the fermentation process may yield functional substances such as antihypertensive peptides. These effects can be considered as “additional benefits” of the matrix, beyond the impact of matrix on probiotic survival both in the product and in your body.
The body of scientific evidence falls short, however, of proving the importance of matrix on health endpoints. For a given amount of probiotics delivered, we lack comparative studies that prove that the end-benefit of one carrier matrix is better than another. Many supportive studies suggest that this will be the case, but until head-to-head human studies are conducted, we don’t know for sure.
Given the impact the matrix exerts on probiotic survival, and the possible effect on probiotic effectiveness, keep in mind the importance of efficacy studies conducted on the complete probiotic product. We need more research to fully understand the role of matrix on probiotic effectiveness, but the strongest evidence comes from studies conducted on the complete probiotic product.
Figure: Determinants of probiotic product parameters (adapted from Flach et al. 2017). Mark B. van der Waal is gratefully acknowledged for producing the artwork.

For another perspective see Does the delivery format affect probiotic efficacy?, March 28, 2018 by Mary Ellen Sanders.