Should bacteriophages be considered as a member of the biotic family?
By Prof. Colin Hill PhD DSc, University College Cork, Ireland
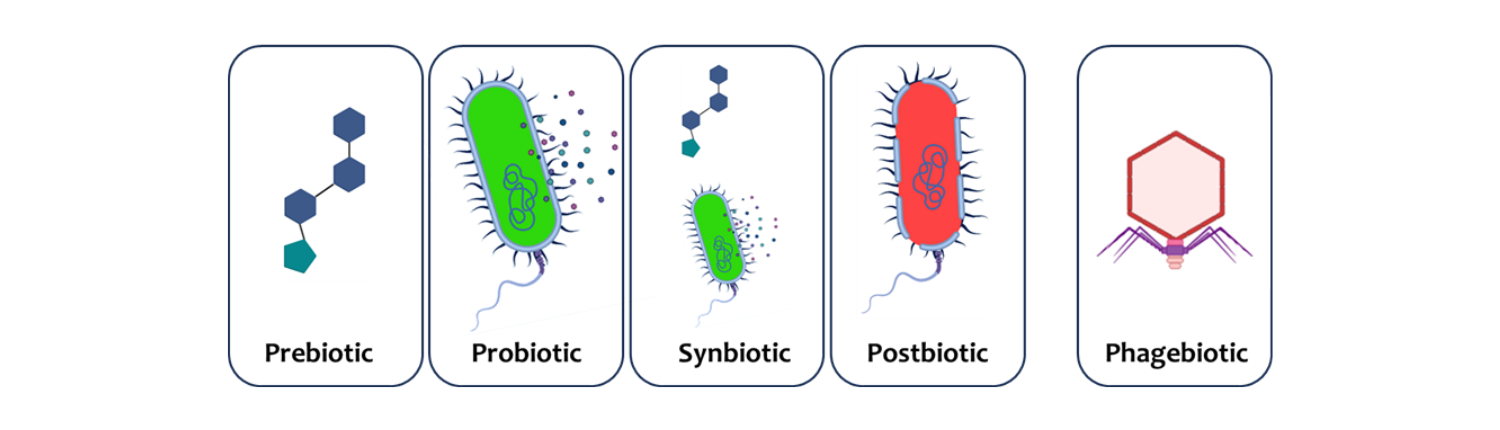
ISAPP has provided consensus definitions for a number of biotics that confer a health benefit on the host. These include prebiotics, probiotics, synbiotics and postbiotics, but here I want to put forward an argument that bacteriophages (phages) could qualify as a new member of the ‘biotic’ family.
Phages are bacterial viruses that infect and replicate within their bacterial victim before bursting the cell and releasing many new copies of the original virus. Phages can also integrate into the bacterial chromosome and co-exist with the living bacterium, but always with the threat that it can excise and initiate another replication-and-burst cycle. Phages are probably the most abundant biological entities on earth and are found wherever bacteria are present in the body. They are an important component of the microbiome of humans, plants and animals, and play a role in regulating bacterial community composition and function.
If phages are to fit neatly within the existing biotic family they would have to qualify as a biotic and also be shown to provide health benefits. The Oxford English Dictionary defines biotics as ‘of or relating to living organisms; caused by living organisms’. Bacteriophages (phages) are not considered as living organisms in themselves, but they easily fit within the biotic definition as they are completely dependent on living bacterial cells for their own propagation and as such certainly ‘relate to living organisms’.
There is also a significant body of evidence that some phages can confer health benefits on a host. Most of this evidence is based on using phage therapy to treat bacterial infections. This has been done in Russia for almost a century, and while the evidence may not always conform to western regulatory standards there is little doubt that phages can bring benefits such as limiting or clearing infections at various body sites. In a recent example, a randomised, controlled, blinded trial on burn wounds was conducted in Belgium and France with Pseudomonas aeruginosa as the target (1). A topically applied preparation consisting of low titres of a 12-phage cocktail was used. While the efficacy did not reach that of the standard-of-care sulfadiazine silver emulsion cream treatment, the phage treatments did lead to sustained reductions in bacterial burdens.
Phages can also be potentially used to modulate microbiomes to impact host health, as shown in a recent study I was involved in performed by Nate Ritz in the John Cryan lab where faecal virome transplants (FVT) changed the bacterial community and thus reduced the impact of stress-induced changes in behaviour and immune responses in mice (2). This paper was the topic of a recent ISAPP podcast for anyone interested in hearing more about that story. FVT has also been reported to work against Clostridioides difficile infections in humans in a small trial in Germany (3).
The term phagebiotic is perhaps the most fitting for this new type of biotic. I have always argued that we should not invent new terms for things that already have names, so why not just stick to bacteriophages or phages? It is because the term phagebiotic would be reserved for a very specific sub-category of phages. Just as all probiotics are microbes, but not all microbes are probiotics, I would suggest that phagebiotics should only be used to refer to specific phage preparations that have been shown to convincingly confer a health benefit in an appropriate properly controlled trial.
Mirroring the probiotics definition I would start with a suggested definition something like this; ‘phagebiotics are bacteriophages that, when administered in adequate amounts, confer a health benefit on the host’.
- Jault P., Leclerc T., Jennes S., Pirnay J.P., Que Y.A., Resch G., Rousseau A.F., Ravat F., Carsin H., Le F.R., et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind phase 1/2 trial. Lancet Infect. Dis. 2019;19:35–45. doi: 10.1016/S1473-3099(18)30482-1
- Ritz, N.L., Draper, L.A., Bastiaanssen, T.F.S. et al. The gut virome is associated with stress-induced changes in behaviour and immune responses in mice. Nat Microbiol 9, 359–376 (2024). https://doi.org/10.1038/s41564-023-01564-y
- Ott, S. J., Waetzig, G. H., Rehman, A., Moltzau-Anderson, J., Bharti, R., Grasis, J. A., et al. (2017). Efficacy of sterile fecal filtrate transfer for treating patients with Clostridium difficile Gastroenterology 152, 799.e797–811.e797. doi: 10.1053/j.gastro.2016.11.010



